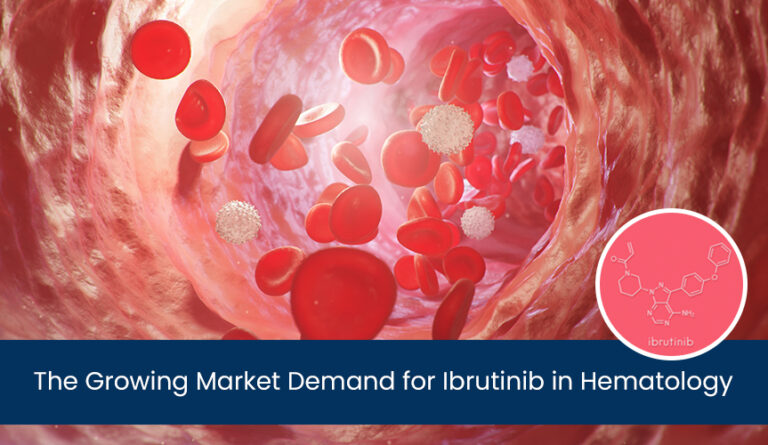
Tofacitinib API is a pharmaceutically active ingredient widely used in treating various autoimmune diseases, including rheumatoid arthritis. Composed of tofacitinib, a Janus kinase (JAK) inhibitor, it helps regulate the immune response by targeting specific signalling pathways. Approved by regulatory authorities, such as the FDA, for multiple indications, including rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis, the Tofacitinib API market has demonstrated efficacy in managing these conditions. Recent developments have expanded its applications, showcasing its potential in treating additional diseases like ankylosing spondylitis and moderate-to-severe atopic dermatitis.
Table of Contents
Overview of Tofacitinib API
Tofacitinib API is a pharmaceutically active ingredient that is widely used in the treatment of autoimmune diseases. It is composed of tofacitinib, a Janus kinase (JAK) inhibitor. This compound targets specific signalling pathways in the immune system, thereby regulating the immune response. Tofacitinib API has been approved for use in various conditions, including rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis.
In recent years, developments and expansions in the indications for the growth opportunity for Tofacitinib API have occurred. It has shown efficacy in treating ankylosing spondylitis and moderate-to-severe atopic dermatitis, expanding its therapeutic applications. These advancements highlight the versatility and market potential of Tofacitinib API in managing a range of autoimmune conditions.
Chemical composition of Tofacitinib API
The chemical composition of Tofacitinib, the active pharmaceutical ingredient (API) in Tofacitinib API, can be described as follows:
Tofacitinib is chemically known as
3-{(3R,4R)-4-methyl-3-[methyl-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino]pyrrolidin-1-yl}-3-oxo propane nitrile. Its molecular formula is C16H20N6O, and its molecular weight is approximately 312.37 grams per mole.
The compound consists of a pyrrolidinyl pyrimidine core structure, which is the active moiety responsible for its pharmacological effects. It also contains a nitrile group (C≡N) and a propanone moiety (C=O), contributing to its chemical properties and interactions.
Tofacitinib API is typically available as a white to off-white crystalline powder; it is soluble in dimethyl sulfoxide (DMSO) and methanol.
It’s important to note that Tofacitinib API is the isolated active ingredient used in the production of medications, and it may be formulated into different dosage forms according to Pharmaceutical industry trends, such as tablets or capsules, along with other excipients and additives for oral administration.
Current Market Landscape of Tofacitinib API
The current market landscape for the Tofacitinib API industry is characterized by substantial growth and competition. With a market size of several billion dollars, Tofacitinib API has attracted major players in the pharmaceutical industry.
Leading multinational companies and generic drug manufacturers have identified the market potential of Tofacitinib API and have become key stakeholders in its production and distribution.
Recent market trends are a sign of a substantial growth rate for Tofacitinib API. The need for effective treatments for autoimmune diseases, such as rheumatoid arthritis and psoriatic arthritis, has expanded its market. Tofacitinib API has grasped a notable market share, establishing itself as a preferred choice among healthcare professionals for managing these conditions.
However, the market landscape is not without challenges. The introduction of generic versions of Tofacitinib API has increased competition, leading to price pressure and potential market fragmentation.
Additionally, the Rheumatoid arthritis treatment market and emerging JAK inhibitors pose a competitive threat to Tofacitinib API’s market dominance.
To maintain a competitive edge, companies involved in the production and distribution of Tofacitinib API must focus on factors such as product quality, cost-effectiveness, and market access. Strategic marketing efforts and research and development investments are vital to stay ahead in this dynamic market landscape.
Market Potential and Growth Factors
The market size for Tofacitinib API is pledging, driven by multiple factors. The rising plurality of rheumatoid arthritis and corresponding conditions associated with an ageing population forms a growing patient pool that needs effective treatments. Additionally, the expanding scope of Tofacitinib API into new indications, such as ankylosing spondylitis and atopic dermatitis, presents untapped market opportunities. Emerging markets, especially in Asia and Latin America, hold significant growth potential due to improving healthcare infrastructure and increased access to novel therapeutics.
Regulatory Environment
The regulatory landscape plays a crucial role in shaping the market outlook of Tofacitinib API. Recent regulatory approvals for expanded indications have bolstered its market potential. However, regulation changes, such as safety warnings or restrictions, can impact market dynamics. Compliance with the regulatory standards, including Good Manufacturing Practices (GMP) and quality control measures, is paramount for sustained market expansion. Adherence to evolving regulatory requirements ensures product safety, efficacy, and public confidence, enabling manufacturers to navigate the complex landscape effectively.
Challenges and Opportunities
The regulatory landscape of Tofacitinib API holds immense growth potential, it faces certain challenges. Safety concerns and adverse events associated with its use have raised caution among healthcare professionals and patients. Addressing these concerns through continued research, post-marketing surveillance, and education is essential to maintain market confidence. Additionally, introducing generic versions and competition from other JAK inhibitors pose challenges to established market players. However, strategic collaborations, research and development initiatives, and geographic expansion into untapped markets offer growth and market diversification opportunities.
Conclusion
The market potential for Tofacitinib API remains strong; it is driven by increasing demand for effective treatments for autoimmune diseases. Its approved indications, recent developments, and expanding applications position it favourably in the market landscape. To achieve this growth, stakeholders should navigate the regulatory environment effectively, address safety concerns, and capitalize on emerging opportunities. With strategic approaches, including research collaborations and geographic expansion, the future of emerging markets for Tofacitinib API appears promising, catering to the needs of patients worldwide and contributing to advancements in autoimmune disease management.