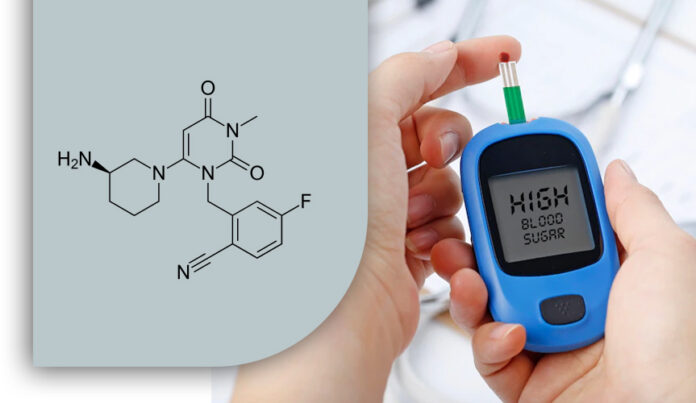
Trelagliptin succinate is a long-acting dipeptidyl peptidase-4 (DPP-4) inhibitor that inhibits DPP-4 activity. Trelagliptin reduces GLP1 breakdown. Thus resulting in a significant reduction in glycated hemoglobin in type 2 diabetes patients.
In addition to its hypoglycemic impact, trelagliptin reduces cholesterol and low-density lipoprotein. It also increases the activity of islet cells in individuals with type 2 diabetes (T2D). A DPP-4 inhibitor combined with insulin decreased insulin resistance in senior T2D patients.
On March 26th, 2015, Trelagliptin succinate, a once-weekly oral dipeptidyl peptidase-4 (DPP-4) inhibitor, got licensed in Japan. Trelagliptin outperformed its analogs alogliptin and sitagliptin.
It was in terms of potency against human DPP-4. And its superior selectivity and slow-binding characteristic may contribute to its long-term effectiveness.
By maintaining certain reaction conditions, you can get a high purity succinate salt of trelagliptin.
Table of Contents
Medical Use Of Trelagliptin Succinate
Trelagliptin inhibits the enzyme dipeptidyl-4 (DPP-4). It also can alleviate insulin resistance in people with type 2 diabetes.
Trelagliptin Succinate – Mechanism Of Action
The pathophysiological foundation of metabolic disorders is insulin resistance. It substantially increases the risk for type 2 diabetes, coronary heart disease, fatty liver, obesity, and hypertension, among other diseases.
Adipose tissue plays a critical function in energy balance by regulating insulin-stimulated glucose intake.
GLUT4, an insulin-sensitive glucose transporter, facilitates glucose transfer in adipose tissue by constantly cycling between intracellular storage vesicles and the plasma membrane.
Any disruption in the translocation of GLUT4 will decrease glucose absorption. And eventually, lead to insulin resistance.
In primary rat adipocytes, trelagliptin succinate increased glucose consumption. Trelagliptin succinate also increased the quantity of GLUT4 protein in the outer membrane. Thus suggesting that trelagliptin succinate stimulated GLUT4 translocation in adipocytes, promoting glucose intake.
In rat adipocytes, trelagliptin succinate reduced the production of free fatty acids and resistin. As a result, the mechanism by which trelagliptin succinate improves insulin resistance might link to a decrease in free fatty acid and resistin production.
It regulates blood glucose levels by inhibiting DPP-4. It is an enzyme inactivating the incretin hormones glucagon-like peptide-1 and glucagon-dependent insulinotropic polypeptide.
These are vital in blood glucose regulation. Inhibition of DPP-4 enhances insulin production in response to blood glucose levels. Thus controlling blood sugar levels.
Synthesis Of Trelagliptin Succinate
The synthesis has gotten much attention because of its unusual therapeutic efficacy and strong market potential.
The original corporate process began with 4-fluoro-2-methylbenzonitrile. It was brominated to produce 2-bromomethyl-4-fluorobenzonitrile and the dibromide 3′ and was subsequently transformed to compound 3 in the presence of diethyl phosphite.
The compound got condensed with 3-methyl-6-chlorouracil to get the crucial intermediate 4. It was subsequently nucleophilically substituted with unprotected (R) -3-aminopiperidine. These reaction conditions yielded trelagliptin.
The nucleophilic substitution is crucial in the synthesis to achieve high regioselectivity. Zhang et al. reported replacing the substance with (R)-3-N-protected-aminopiperidine to avoid the creation of the regio-isomer in 2016. However, the subsequent deprotection procedure may yield additional impurities due to cyano-hydrolysis.
Trelagliptin succinate resulted from dissolving crude and succinic acid in isopropyl alcohol. Then recrystallising it using a 9:1 combination of isopropyl alcohol and water. Thus, giving a high purity succinate salt of Trelagliptin.
Side Effects Of Trelagliptin Succinate
It may lead to hypoglycemia (0.15%). In such cases, provide medicine while carefully monitoring the patient’s condition. Other DPP-4 inhibitors link to significant hypoglycemia and even loss of consciousness when used with Sulfonylurea medicines.
Furthermore, if low blood sugar happens due to taking this drug, it can get treated with sucrose. Hypoglycemia caused by glucosidase inhibitors, on the other hand, should be treated with fructose.
Drug Approval
Takeda pharmaceutical company submitted a new drug application for trelagliptin to treat T2DM in Japan in March 2014.
The Japanese Ministry of Health (MHLW) approved the medicine in March 2015. It got approved based on the favourable outcomes of phase III studies in T2DM patients.
Trelagliptin had completed phase II trials outside of Japan. However, Takeda decided to stop developing it in the United States and the European Union in October 2014. It was due to the high expenses of getting permission in these countries.
Precautions
People with the following health conditions should be careful while administering the drug. That’s because it may lead to further complications.
- Mild renal function problem.
- People on sulfonylurea or insulin therapy. There have been instances of significant hypotension when used with other DPP-4 inhibitors.
- Hypopituitarism is also known as hypoadrenalism. It is a health condition wherein the pituitary gland is underactive.
- Malnutrition, malnutrition, irregular eating patterns, or hypoesthesia are all possible causes of hypoesthesia.
- People who exercise vigorously.
- Patients who drink too much alcohol.
Conclusion
Trelagliptin is a succinate salt that has given promising results in reducing insulin resistance, thus alleviating type 2 diabetes. Trelagliptin succinate gained approval in March 2015 In Japan. Even with successful results, you still need to be cautious while administering the drug.
For the best quality active pharmaceutical ingredient company, you can check out our website or call us if you have any queries.